Role of Spectral Line Physics in CO₂ Retrieval
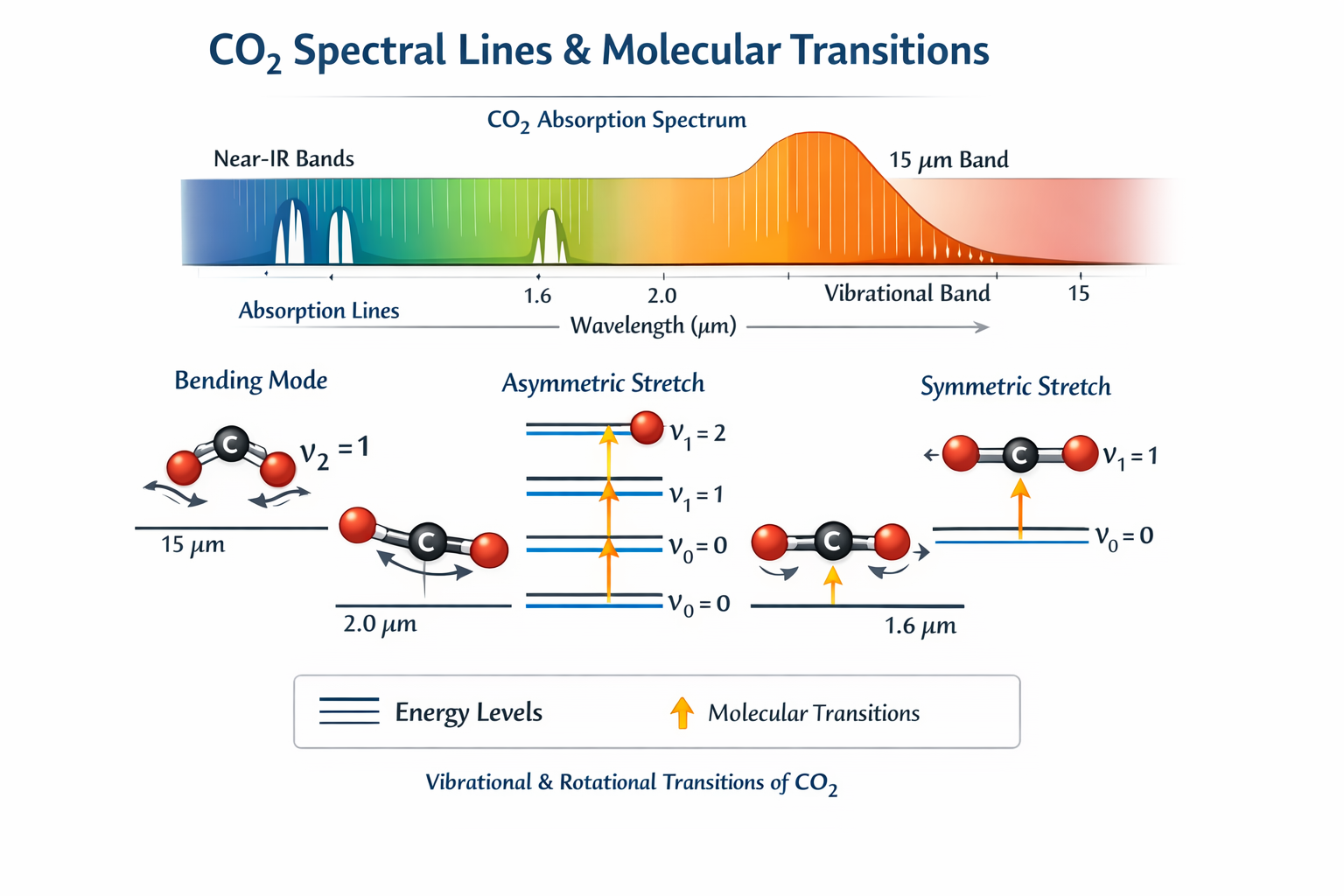
Physical Origin of Spectral Lines
CO₂ molecules absorb radiation when photons match the energy difference between molecular quantum states. For vibrational–rotational transitions: $$\Delta E = h \nu_0$$ Thus absorption occurs at discrete frequencies \( \nu_0 \).In SWIR bands \(\sim 1.6 \mu m, \sim 2.0 \mu m\), these correspond to overtone and combination vibrational bands of CO₂. Each transition produces a spectral line. The observed absorption band is a superposition of thousands of individual lines.
From Microscopic Transitions to Macroscopic Absorption
Microscopically, absorption probability per molecule: $$ \sigma_\nu $$ Macroscopic absorption coefficient: $$ \alpha_\nu = n \sigma_\nu $$ Where:- \( n \) = number density
- \( \sigma_\nu \) = absorption cross-section
Mathematical Description of a Spectral Line
For a single transition: $$ \sigma_\nu = S(T) f(\nu - \nu_0) $$ Where:- \( S(T) \) = line strength
- \( f(\nu - \nu_0) \) = line shape function
- \( \nu_0 \) = line center
Line Strength \( S(T) \)
Line strength depends on temperature via Boltzmann population: $$ S(T) = S(T_0) \frac{Q(T_0)}{Q(T)} \exp\left( -\frac{E_l}{k_B} \left( \frac{1}{T} - \frac{1}{T_0} \right) \right) $$ Where:- \( Q(T) \) = partition function
- \( E_l \) = lower state energy
Line Shape Physics
Real lines are broadened by physical mechanisms.- Doppler Broadening:
Due to molecular thermal motion.
Velocity distribution → frequency shift.
Width: $$ \alpha_D =\nu_0 \sqrt{\frac{2k_B T}{mc^2}} $$ Line shape: $$ f_D(\nu) = \frac{1}{\alpha_D \sqrt{\pi}} \exp\left( -\frac{(\nu - \nu_0)^2}{\alpha_D^2}\right) $$ Dominates in upper atmosphere (low pressure). - Pressure (Collisional) Broadening
Molecular collisions perturb energy levels.
Lorentzian profile:
$$ f_L(\nu) = \frac{1}{\pi} \frac{\gamma}{ (\nu - \nu_0)^2 + \gamma^2} $$ Where: $$ \gamma = \gamma_{air} p_{air} + \gamma_{self} p_{CO_2} $$ Dominates in lower troposphere. - Voigt Profile: Real atmosphere → convolution of Doppler and Lorentz: $$ f_V(\nu) = f_D * f_L $$ This profile determines exact absorption line shape.
From Spectral Lines to Optical Depth
Gas optical depth: $$ \tau_\nu^{CO_2} = \int_0^\infty n_{CO_2}(z) \sigma_\nu^{CO_2}(T,p) dz $$ But: $$ \sigma_\nu^{CO_2} = \sum_i S_i(T) f_i(\nu - \nu_i, p, T) $$ Thus: $$ \tau_\nu = \sum_i \int n(z) S_i(T(z)) f_i(\nu - \nu_i, p(z), T(z)) dz $$ This equation shows:- Line strengths weighted by temperature
- Line shapes weighted by pressure
- Integrated over vertical column
Why Spectral Line Physics Is Critical
Retrieval does not simply measure total absorption. It fits:- Line depth
- Line width
- Line wings
- Line asymmetry
- CO₂ amount
- Pressure profile
- Temperature
- Path length
How Retrieval Algorithms Use Spectral Line Physics
Forward model workflow:- Read line parameters from database (e.g. HITRAN)
- Line center
- Line strength
- Air broadening coefficients
- Temperature exponents
- Compute line strength at actual temperature.
- Compute Voigt profile at each altitude.
- Compute absorption cross-section: $$ \sigma_\nu(z) = \sum_i S_i(T(z)) f_i(\nu, p(z), T(z)) $$
- Compute vertical optical depth: $$ \tau_\nu = \int n_{CO_2}(z) \sigma_\nu(z) dz $$
- Insert into forward radiance model: $$ I_\nu = I_0 e^{-\tau_\nu} $$
- Convolve with instrument line shape.
How Spectroscopy Errors Cause Bias
Suppose line strength has error: $$ S = S_{true} + \delta S $$ Optical depth becomes: $$ \tau_\nu \propto S $$ Measured absorption depth fixed. Thus retrieval compensates by adjusting CO₂: $$ \delta XCO_2 \approx -\frac{\delta S}{S} XCO_2 $$ 1% line strength error → ~1% CO₂ bias (~4 ppm). That is mission-critical.Subtle Spectroscopic Effects
Advanced retrievals must also include:- Line mixing
- Speed dependence
- Non-Voigt corrections
- CIA (collision-induced absorption)
- Temperature-dependent broadening
Conceptual Summary
Spectral line physics defines: $$\sigma_\nu \rightarrow \tau_\nu \rightarrow I_\nu \Rightarrow XCO_2$$ Thus spectroscopy is the physical bridge between quantum mechanics and climate science.Reference
- Fundamentals of Remote sensing
- Relevance of Electromagnetic waves in the context of earth observation
- Concept of the orbits for a satellite (non scientific discussion)
- How various teams works in close collaboration for the ground data processing?
- How raw satellite data is processed to do a level where you do your scientifc research?
- In depth understandingof the satellite data (op-of-atmosphere reflectance)
- Resolution and calibration
- Understanding how OLCI data is processed
- Transforming Energy into Imagery: How Satellite Data Becomes Stunning Views of Earth
